 In which U coul and U van are the coulomb energy and the van der Waals energy extracted from the MD simulations. Here, the size effect on the viscosity of the confined water implies the influence of the diameter of SWCNTs. The objective of this study is to examine the size and the temperature effects on the water viscosity. 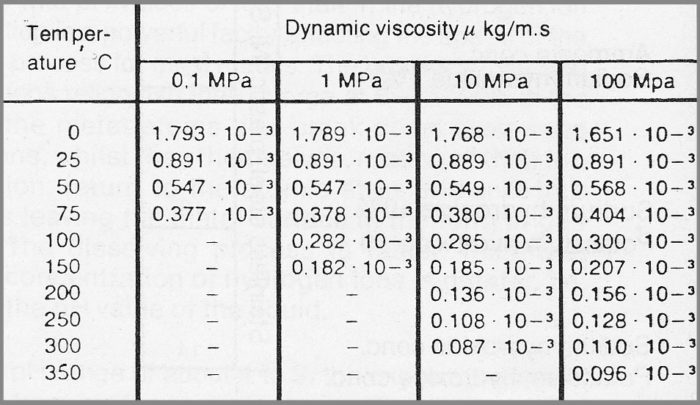
In this article, we redetermine the coefficients in the "Eyring-MD" method through more numerical experiments and evaluate the viscosity of water inside SWCNTs at 298, 325, and 350 K. Recently, an "Eyring-MD" method was proposed to calculate the viscosity of water by using the MD simulations. This restricts the application of the classical continuum theory to the nanoflows. So far, the viscosity of fluids in nanoconfinement on a scale comparable to the molecular diameter is seldom explored owing to the extremely small scale on which the transport properties are difficult to be captured by experiments and the intrinsic limitations of the existing computational methods in the MD simulations. The previous results have identified that the water viscosity relies on the temperature and the characteristic length of the nanochannel. In classical continuum theory, the viscosity is an essential transport property and thereby has been extensively measured and computed. Hence, many researches focused on the unique feature of the confined fluid and its relationship with the continuum fluid. The previous studies have revealed that the flow behavior of water at the nanoscale strongly depends on the characteristic length of nanochannel, which implies that the classical continuum theory for the macroscopic fluid may be no longer applicable for the fluid confined in nanochannels. It is a significant topic for studying and designing the nanodevices such as the nanochannel for drug delivery and the membrane for water desalination. Water conduction through single-walled carbon nanotubes (SWCNTs) has been paid much attention in recent years. The present results should be instructive for understanding the coupling effect of the size and the temperature at the nanoscale. The results of the relative amount of the hydrogen bonds exhibit similar profiles with the curves of the relative viscosity. To demonstrate the rationality of the calculated relative viscosity, the relative amount of the hydrogen bonds of water confined in SWCNTs is also computed. Based on the computational results, a fitting formula is proposed to calculate the size- and temperature- dependent water viscosity, which is useful for the computation on the nanoflow. The results suggest that the relative viscosity of the confined water increases with increasing diameter and temperature, whereas the size-dependent trend of the relative viscosity is almost independent of the temperature. This does not yet include AV's latest tweaks (M3 above) - however the differences for most applications should be quite minor.The influences of the diameter (size) of single-walled carbon nanotubes (SWCNTs) and the temperature on the viscosity of water confined in SWCNTs are investigated by an "Eyring-MD" (molecular dynamics) method. The other (written by A.V.) takes theĪ python version has now also been written by Matthew Partridge at Cranfield University. One takes volume fraction of glycerine as input. 
Further refinements from Andreas Volk to density of pure water, and the temperature-dependence of the contraction of the mixture. In both cases the fit was chosen to match data from Gregory (table 3 and 7) Andreas Volk pointed out that the density calculation can be made more accurate by (i) accounting for the volume contraction of the mixture (ii) adjusting the fit for the density of pure glycerine as a function of temperature. Thanks to Paul Debue for pointing this out. The mixture should use the glycerine fraction by VOLUME and not by mass. Density calculation has been changed: equation 25 in Cheng's paper to compute the density of I'd recommend reading the latter paper first. Kähler (2018) Experiments in Fluids 59 75. 47 3285-3288, with a number of adjustments (see below), which are described in Volk and The calculation is based on the parameterisation in Cheng (2008) Ind. Calculate density and viscosity of glycerol/water mixturesĭynamic viscosity of mixture is : 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
